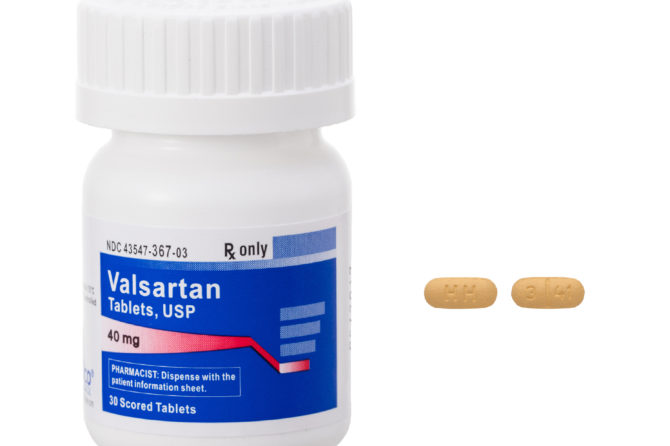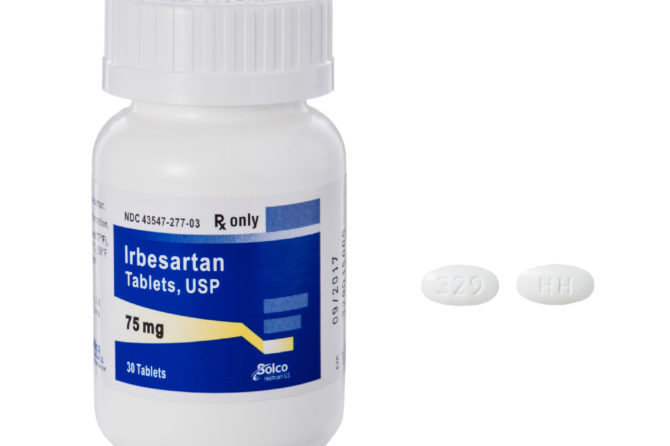
Prinston Pharmaceutical Inc. issues Voluntary Nationwide Recall of Irbesartan and Irbesartan HCTZ Tablets Due to detection of a Trace Amount of Unexpected Impurity, Nnitrosodiethylamine (NDEA) in the Products
Read PDF here. http://www.solcohealthcare.com/uploads/news/Press-Release-Irbesartan-Recall.pdf
Read more